Quorum sensing is the ability of single-celled organisms to measure the cell density of the population via chemical communication.
| An essential part of your facility is the Biofilter. This determines the success of your system. In this context, the properties of the bacteria used for the purification, conversion and treatment of your nutrients are of great importance. This article explores how bacteria communicate with each other. If the environmental conditions for your filter aren't optimally adjusted, it may simply not work at all in the worst case, or it may only be able to operate at a fraction of its capacity in the slightly cheaper case. According to fisheries experts from the LANUV and the Ministry's experience from practice is that new plants produce only about 10% - 30% of the maximum possible biomass in the first few years. In stable operation, recirculation systems are operated at approximately 70% - 80% of their capacity. |
Quorum sensing (QS) is the ability of single-celled organisms to measure the cell density of their own species' population and the complexity of the community via chemical communication using highly specific signaling molecules. Contact of the bacteria with higher organisms is also regulated via QS. Bacteria can respond quickly to changes in their environment via QS to ensure population survival, gain advantages over competitors, and develop new suitable ecological niches. In doing so, they can specifically disrupt the communication of other bacteria. The QS system allows microorganisms to reproduce protected in the community, in biofilms, up to a critical cell number, and then change the population phenotype together. Genes are only activated ifwhen a certain cell density, the quorum, is exceeded or undercut.
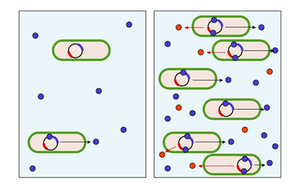 |
Scheme of quorum sensing,left: concentration of autoinductor molecules (blue) low,right: concentration of autoinductor molecules high, thus synthesis of the bacterial product (red). Y_tambe • CC-BY-SA-3.0 |
In itself, the term „quorum“ dates back to the time of the Roman Empire and referred to the smallest number of members required for a vote in the Senate.
Quorum sensing is used by bacteria to coordinate processes that would be inefficient if performed only by individual cells, e.g. B. Bioluminescence, biofilm formation, secretion of antibiotics and pathogenicity factors, fruiting body formation in myxobacteria, sporulation in Bacillus subtilis, infection of plants[3] and other eukaryotic hosts[4], and bacterial involvement in ecosystems such as coral reefs.[5] QS coordinates the behavior of a species' bacteria in a very small space. Pseudomonas aeruginosa, a causative agent of pneumonia and other infections, can live within its host without harming it. However, when it multiplies rapidly, the cells form biofilms, become pathogenic, and can lead to host disease.[6]
The importance of autoinductors in QA
The autoinductors are generally freely diffusing, amphiphilic molecules that are constantly released into the environment by the bacteria in small amounts via the cell membrane. In Gram-negative bacteria, these are predominantly small molecule compounds, while in Gram-positive bacteria they are oligopeptides.[10] Through the concentration of the delivered signaling molecules, bacteria can measure population density and community complexity and, above a critical concentration, change the phenotype of the population. In complex environments, bacteria can communicate simultaneously with different autoinducers via different QS systems and adapt collectively to the respective situation. This gives them a competitive advantage over other bacteria.[11] Thus, the marine Vibrio harveyi uses three different autoinducers for interspecies communication and exchange between genera. In total, up to 600 genes can be regulated.[12][13]
The secreted autoinducers can be assigned to different groups based on their chemical structure and receptors:
Autoinductor-1 (Al-1)
Autoinductor-1 (Al-1) is mainly present in Gram-negative bacteria and serves exclusively for intraspecific communication. Most bacteria use acylated homoserine lactones (N-acyl homoserine lactone, (AHL)) as a signaling molecule, some also use aryl homoserine lactone for interspecific communication. The synthesis starts from the amino acid S-adenosylmethionine. The length of the alkyl chain of N-acyl homoserine lactone can vary, exhibit modifications, and thereby additionally influence the stability and signaling dynamics of the molecule.[14][15] The N-acyl homoserine lactones are produced using bacterial enzymes from the LuxI synthase family (AHL synthase).[16] The hydrophobic AHL molecules are membrane-permeable and accumulate in the environment of the bacteria.After reaching the threshold concentration, AHL bind to cytoplasmic receptor proteins of the LuxR family and induce diverse biological processes by binding to DNA.[17] This LuxI/LuxR-QS system with AHL as a signaling molecule is typical for many Gram-negative bacteria.
There are different variants of the system, for example in Pseudomonas aeruginosa with the autoinductor N-butyroyl-L-homoserine lactone and the RhlI/RhlR-QS system and in Chromobacterium violaceum with N-hexanoyl-L-homoserine lactone and the Cvil/CviR-QS system.[18]
Many proteobacteria have additional LuxR homologous receptors in addition to the LuxI/LuxR QS system, but no synthases related to LuxI synthase. These systems are called LuxI solo. They enable bacteria to respond to exogenously produced AHL and thus enter into communication with other non-species bacteria.[19]
In addition to these common AHL, some Gram-negative pathogenic bacteria use other signaling molecules for communication with their hosts, but receptors that are homologous to the LuxR type. For example, the insect pathogenic Photorhabdus luminescens uses 2-pyrone or photopyrone as signaling molecules formed by pyrone synthase (Ppys) that bind to the LuxR homologous receptor PluR. Through this PpyS/PluR-QS system, the pathogen controls the cell clumping associated with virulence.[20] The insect pathogenic and human pathogenic bacterium Photorhabdus asymbiotica also does not possess LuxI synthase. This bacterium controls communication via dialkylresorzinols and cyclohexanediones, which also bind to LuxR homologous PauR receptors and thus regulate virulence via the DarA/DarB/DarC/PauR QS system.[21]
Autoinductor-2 (Al-2)
Autoindutkor-2 (Al-2) occurs in Gram-negative and Gram-positive bacteria of various taxa and is involved in interspecies communication but not universally represented.[22] Chemically, they are cyclic furanosylborate diesters.[23] Starting from the precursor, the 4,5-dihydroxy-2,3-pentanedione (DPD), formed by the LuxS enzyme, DPD decomposes in aqueous solution into two enantiomers that are in chemical equilibrium. 4,5-Dihydroxy-2,3-Pentanedione complexes with boron to form the cyclic furanosylborate diester and forms the signaling molecule Al-2.[24] The synthesis of Al-2 also appears to be regulated via positive feedback.[25] In Vibrio cholerae, Al-2 binds to the periplasmatically localized receptor LuxP, which interacts with the sensor kinase LuxQ,which can act as a kinase or phosphatase depending on the status of the QS system and thus, for example, switches on or off the expression of the genes for biofilm formation.[26] Al-2 provides information about metabolic activities of the bacteria in the environment and about their microbial colonization. In the bacterial populations of Escherichia coli and Salmonella typhimurium, environmental conditions: preferred carbon sources, low pH and high osmolarity, induce Al-2 synthesis, while poor conditions, stationary growth phase of the bacterial population, depleted carbon sources, low osmolarity and altered pH, induce Al-2 degradation.[27][26] Al-2 provides information about metabolic activities of the bacteria in the environment and about their microbial colonization. In the bacterial populations of Escherichia coli and Salmonella typhimurium, environmental conditions: preferred carbon sources, low pH and high osmolarity, induce Al-2 synthesis, while poor conditions, stationary growth phase of the bacterial population, depleted carbon sources, low osmolarity and altered pH, induce Al-2 degradation.[27][26] Al-2 provides information about metabolic activities of the bacteria in the environment and about their microbial colonization. In the bacterial populations of Escherichia coli and Salmonella typhimurium, environmental conditions: preferred carbon sources, low pH and high osmolarity, induce Al-2 synthesis, while poor conditions, stationary growth phase of the bacterial population, depleted carbon sources, low osmolarity and altered pH, induce Al-2 degradation.[27]an induction of the synthesis of Al-2, while poor conditions, stationary growth phase of the bacterial population, depleted carbon sources, low osmolarity and altered pH result in degradation of Al-2.[27]an induction of the synthesis of Al-2, while poor conditions, stationary growth phase of the bacterial population, depleted carbon sources, low osmolarity and altered pH result in degradation of Al-2.[27]
Autoinductor-3 (Al-3)
Autoinductors-3 (Al-3), in addition to other autoinductors, are found in various pathogenic Gram-negative bacteria such as enterohemorrhagic Escherichia coli (EHEC), vibrios and in Gram-positive pathogens such as Staphylococcus aureus and are induced under stress conditions. Chemically, Vibrio cholerae are various pyrazine metabolites, such as 3,5-dimethylpyrazin-2-ol (DPO), which are formed from the amino acid L-threonine by threonine dehydrogenase. DPO binds to the cytoplasmic LuxR receptor VqmA. This signal-receptor complex induces the transcription of VqmR of an sRNA that can regulate multiple mRNAs from different QS systems. While VqmA controls the transcription factors for biofilm binding,At the same time, the transcription factors for the virulence factor genes can be inhibited by VqmA.[28] The autoinductors 2,5-dimethylpyrazine (DMP) and 3,5-disubstituted pyrazin-2-ol analogue are synthesized from the precursor aminoacetone.[29]
Autoinductor peptides
The Gram-positive bacteria use linear or cyclic oligopeptides, autoinductor peptides. This very heterogeneous group of peptides are synthesized as prepropeptides and activated during active transport. Examples of this group include the peptide hormone or autoinducing peptide (AIP) of Staphylococcus aureus and the competence-stimulating peptide (CSP) of Streptococcus mitis and Streptococcus pneumoniae.[30] In Staphylococcus aureus, AIP binds to the QS receptor AgrC, a transmembrane receptor with a histidine kinase that activates an intracellular transcription factor through phosphorylation and thus induces gene expression. The signal is transmitted here via a two-component system in the cell. The four components of the QS system are genetically organized in an arg operon.[31]
Sources:
- Kenneth H. Nealson, Terry Platt and J. Woodland Hastings: Cellular control of the synthesis and activity of the bacterial luminescent system. In: Journal of Bacteriology, Volume 104, Number 1, p. 313-22, 1970, doi:10.1128/jb.104.1.313-322.1970, PMID 5473898, PMC 248216 (free full text).
- ↑ WC Fuqua, SC Winans, EP Greenberg: Quorum sensing in bacteria: the LuxR-LuxI family of cell density-responsive transcriptional regulators. In: Journal of bacteriology. Volume 176, Number 2, January 1994, pp. 269–275, PMID 8288518, PMC 205046 (free full text), (review).
- ↑ Kathrin Riedel, Susan Schönmann and Leo Eberl: Quorum sensing in plant-associated bacteria. In: BIOspectrum, Volume 11, Year 4, pp. 385–388, (free full text).
- ↑ Bonnie L. Bassler and Richard Losick: Bacterially speaking. In: Cell (magazine), Volume 125, 2nd year, p. 237–246, April 21, 2006, doi:10.1016/j.cell.2006.04.001, (free full text).
- ↑ Laura R. Hmelo: Quorum Sensing in Marine Microbial Environments. In: Annual Review of Marine Science, Volume 9, p. 257–281, January 3, 2017, doi:10.1146/annurev-marine-010816-060656.
- ↑ Roger S. Smith and Barbara H. Iglewski: Pseudomonas aeruginosa quorum sensing as a potential antimicrobial target. In: Journal of Clinical Investigation, Volume 112, Number 10, p. 1460–1465, November 15, 2003, doi:10.1172/JCI20364, (free full text).
- ↑ Franziska S. Birmes and Susanne Fetzner: Quorum sensing Bacterial communication: signals and signal-inactivating enzymes. In: BIOspectrum, 22nd year, 2016, doi:10.1007/s12268-016-0681-4, (free full text).
- ↑ Michael J. Federle: Autoinducer-2-based chemical communication in bacteria: complexes of interspecies signaling. In: Contributions to Microbiology, Volume 16, Number 18, 2009, PMID 19494577, doi:10.1159/000219371.
- ↑ H. Sztaljer, A. Lemme and I. Wagner-Döbler: Quorum sensing and caries. In: BIOspectrum, 14th year, pp. 578–582, 2008, (free full text).
- ↑ Yannick Hecher and Kai Papernfort: Small, dangerous and talkative – quorum sensing in Vibrio cholerae. In: BIOspectrum, Volume 26, Issue 2, p. 136–138, March 2020, doi:10.1007/s12268-020-1344-z.
- ↑ Yannick Hecher and Kai Papenport: Small, dangerous and talkative – quorum sensing in Vibrio cholerae. In: BIOspectrum, Volume 26, Number 2, p. 136–138, March 2020, doi:10.1007/s12268-020-1344-z.
- ↑ Melissa B Miller, Karen Skorupski, Derrick H Lenz, Ronald K Taylor and Bonnie L Bassler: Parallel quorum sensing systems convert to regulate virulence in Vibrio cholerae. In: Cell (magazine), Volume 110, Issue 3, p. 303-14, August 9, 2002, doi:10.1371/journal.ppat.1008313, (free full text).
- ↑ Julia C van Kessel, Steven T Rutherford, Yi Shao, Alan F Utria and Bonnie L Bassler: Individual and combined roles of the master regulators AphA and LuxR in control of the Vibrio harveyi quorum-sensing regulon. In: Journal of Bacteriology, Volume 195, Issue 3, p. 436-43, February 2013, doi:10.1128/JB.01998-12, (free full text).
- ↑ Kai Papenfort and Bonnie Bassler: Quorum-Sensing Signal-Response Systems in Gram-Negative Bacteria. In: Nature Reviews Microbiology, Volume 14, Number 9, p. 576–588, August 11, 2016, doi:10.1038/nrmicro.2016.89, PMID 27510864.
- ↑ Warren RJD Galloway, James T. Hodgkinson, Steven D. Bowden, Martin Welch and David R. Spring: Quorum sensing in Gram-negative bacteria: small-molecule modulation of AHL and AI-2 quorum sensing pathways. In: Chemical Reviews, Volume 111, Number 1, p. 28–67, January 12, 2011, doi:10.1021/cr100109t.
- ↑ Rebecca J Case, Maurizio Labbate and Staffan Kjelleberg: AHL-driven quorum-sensing circuits: their frequency and function among the Proteobacteria. In: ISME Journal, Volume 2, Number 4, p. 345-9, April 2008, doi:10.1038/ismej.2008.13.
- ↑ Kai Papenfort and Bonnie Bassler: Quorum-Sensing Signal-Response Systems in Gram-Negative Bacteria. In: Nature Reviews Microbiology, Volume 14, Number 9, p. 576–588, August 11, 2016, doi:10.1038/nrmicro.2016.89, PMID 27510864.
- ↑ Nazzareno Dominelli and Ralf Heermann: The silent communication of the bacteria Small Talk. In: Biology in our time, Volume 50, p. 414–423, 2020, doi:10.1002/biuz.202010720, (free full text).
- ↑ Sophie Brameyer, Darko Kresovic, Helge B Bode and Ralf Heermann: Dialkylresorcinols as bacterial signaling molecules. In: Proceedings of the National Academy of Sciences, Volume 112, Number 2, p. 572-7, 2015, doi:10.1073/pnas.1417685112, (free full text).
- ↑ Nazzareno Dominelli and Ralf Heermann: The silent communication of the bacteria Small Talk. In: Biology in our time, Volume 50, p. 414–423, 2020, doi:10.1002/biuz.202010720, (free full text).
- ↑ Sophie Brameyer, Darko Kresovic, Helge B Bode and Ralf Heermann: Dialkylresorcinols as bacterial signaling molecules. In: Proceedings of the National Academy of Sciences, Volume 112, Number 2, p. 572-7, 2015, doi:10.1073/pnas.1417685112, (free full text).
- ↑ Michael J. Federle: Autoinducer-2-based chemical communication in bacteria: complexes of interspecies signaling. In: Contributions to Microbiology, Volume 16, p. 18–32, 2009, doi:10.1159/000219371, PMID 19494577.
- ↑ Michael J. Federle. Autoinducer-2-based chemical communication in bacteria: complexes of interspecies signaling. In: Contributions to Microbiology, Volume 16, p. 18–32, June 2, 2009, doi:10.1159/000219371, PMID 19494577.
- ↑ Helena Sztajer, André Lemme and Irene Wagner-Döbler: Streptococcus mutans quorum sensing and caries. In: BIOspectrum, 14th year, 2008.
- ↑ Helena Sztajer, André Lemme, Ramiro Vilchez, Stefan Schulz, Robert Geffers, Cindy Ying Yin Yip, Celine M. Levesque, Dennis G. Cvitkovitch, and Irene Wagner-Döbler: Autoinducer-2-regulated genes in Streptococcus mutans UA159 and global metabolic effect of the luxS mutation. In: Journal of Bacteriology, Volume 190, Number 1, p. 401–415, 2008, doi:10.1128/JB.01086-07, PMID 17981981, (free full text).
- ↑ Yannick Hecher and Kai Papenfort: Small, dangerous and talkative – quorum sensing in Vibrio cholerae. In: BIOspectrum, Volume 26, Number 2, p. 136–138, 2020, doi:10.1007/s12268-020-1344-z.
- ↑ Michael G. Surette, Melissa B. Miller, and Bonnie L. Bassler: Quorum sensing in Escherichia coli, Salmonella typhimurium, and Vibrio harveyi: A new family of genes responsible for autoinducer production. In: Proceedings of the National Academy of Sciences, Volume 96, Number 4, February 16, 1999 p. 1639–1644, doi:10.1073/pnas.96.4.1639, (free full text).
- ↑ Yannick Hecher and Kai Papenfort: Small, dangerous and talkative – quorum sensing in Vibrio cholerae. In: BIOspectrum, Volume 26, Number 2, p. 136–138, 2020, doi:10.1007/s12268-020-1344-z.
- ↑ Chung Sub Kim, Alexandra Gatsios, Santiago Cuesta, Yick Chong Lam, Zheng Wei, Haiwei Chen, Regan M. Russell, Emilee E. Shine, Rurun Wang, Thomas P. Wyche, Grazia Piizzi, Richard A. Flavell, Noah W. Palm, Vanessa Sperandio and Jason M. Crawford: Characterization of Autoinducer-3 Structure and Biosynthesis in E. coli. In: ACS Central Science, Volume 6, Number 2, p. 197–206, January 22, 2020, doi:10.1021/acscentsci.9b01076, PMID 32123737, PMC 7047286 (free full text).
- ↑ Frederick Verbeke, Severine De Craemer, Nathan Debunne, Yorick Janssens, Evelien Wynendaele, Christophe Van de Wiele and Bart De Spiegeleer: Peptides as Quorum Sensing Molecules: Measurement Techniques and Obtained Levels In vitro and In vivo. In: Frontiers in Neuroscience, Volume 11, p. 183, April 12, 2017, doi:10.3389/fnins.2017.00183, (free full text).
- ↑ Joseph K. Vasquez and Helen E. Blackwell: Simplified Autoinducing Peptide Mimetics with Single-Nanomolar Activity Against the Staphylococcus aureus AgrC Quorum Sensing Receptor. In: ACS Infectious Diseases, Volume 5, Number 4, p. 484–492, 2019, doi:10.1021/acsinfecdis.9b00002, (free full text).
- https://de.m.wikipedia.org/wiki/Quorum_sensing
Image: Schematic of quorum sensing,left: concentration of autoinductor molecules (blue) low,right: concentration of autoinductor molecules high, thus synthesis of the bacterial product (red). Y_tambe • CC-BY-SA-3.0


Add Comment